About
LuroMed LLC develops and commercializes proprietary polymer technology for use in therapeutic topical skin care and in medical applications including soft tissue augmentation and drug delivery. In the skincare field, the company's patented technology utilizes natural polysaccharide chemistry to produce biopolymers which bind to, and deliver active ingredients, including water, to the skin.
Vantage Specialty Ingredients - www.lipochemicals.com is the exclusive global distributor for five of LuroMed's HA-based products, including Hylasome®EG10, one of the family of chemically cross-linked HA gels.
The safety and efficacy of these proprietary polymers makes them value-added ingredients for use in high performance products developed to: manage dermal disorders (including dry, inflamed, burned, bruised or injured skin), improve the health and appearance of skin, protect the skin from further injury or damage from environmental stressors and address the damaging effect of ultraviolet radiation on the skin.
LuroMed has developed a line of finished product formulations for the personal care market. LuroMed's proprietary polymer technologies are used in the formulations to provide moisturization, substantivity, and protection to the skin.
In the medical field, the company's novel, patented co-polymeric HA-silicone technology is designed to provide improved stability and efficacy of soft tissue implants and to provide safe, biocompatible, long-lasting depot delivery of actives.HA-Si copolymers are resistant to enzymic degradation (hyaluronidase) as compared to pure crosslinked HA gels, measured by loss in viscosity:
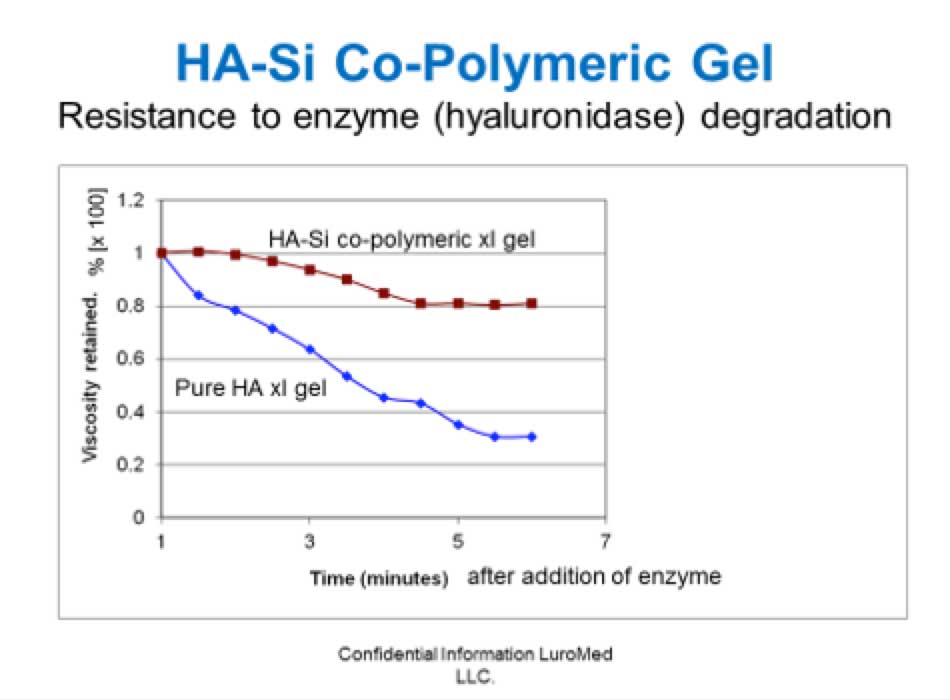
HA-Si co-polymers are heat stable, exhibit hydrophilic and hydrophobic properties, and have customizable physical properties ie soft to firm gels.

LuroMed LLC has developed technology for various HA-based products, including unique cross-linked HA-polymer complexes used in personal care products. LuroMed also develops and produces its own HA ingredients; large scale production is conducted at contract manufacturing facilities in NY and NJ under cosmetic GMP (Good Manufacturing Practices, USFDA).
HA is a useful and effective component in personal care, particularly skin care products due to its natural ability to moisturize, protect (free radical scavenger), heal (wounds) and deliver actives. LuroMed understands the importance of physical and chemical properties of HA (viscoelasticity, molecular weight, concentration, purity) and the need to maintain and control these properties during and after manufacture.
Example:
Hylasome®EG10
INCI: sodium hyaluronate crosspolymer
CAS #: 380600-88-4, 855781-05-4
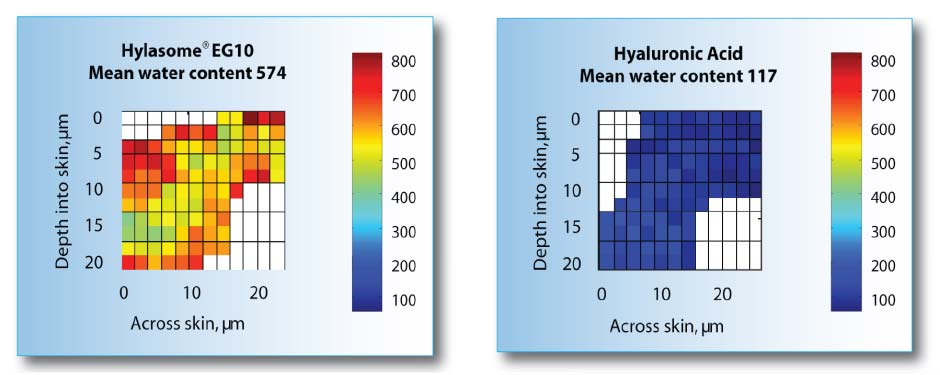
Moisturization of skin: Hylasome®EG10 vs Hyaluronic Acid